Gemini™
608 miles on a single charge

Specifications
Energy
185+ kWhGED
260 Wh/kgVED
450 Wh/LCell-to-Pack Efficiency
70%System Configuration
204S3PDimensions
2,640 x 1,490 x 128 mmMass
710 kgVolume
411 LVoltage
748 VPeak Power
345 kWContinuous Power
130 kWCycle Life
>250,000 milesDoubling range safely.
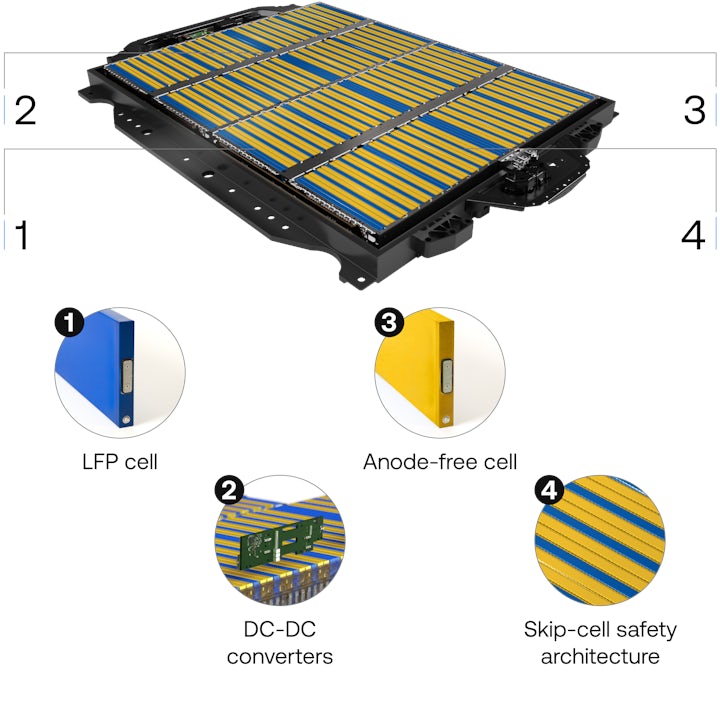
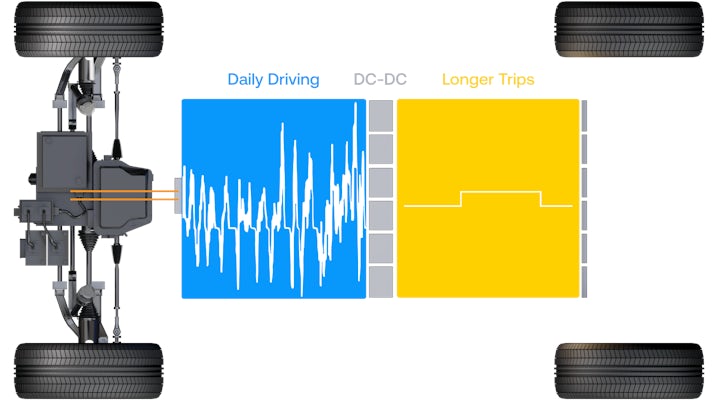
Electric vehicles will go mainstream when people are comfortable with an electric vehicle as their only vehicle. Gemini is an architecture designed to double the range of electric vehicles without compromising safety. It uses two cells: a lithium iron phosphate (LFP) cell for daily driving and an anode-free cell for longer trips. A DC-DC converter moves energy from between them seamlessly while our patented skip-cell architecture sharply reduces the risk of thermal runaway.
Gemini Powers BMW iX 608 Miles
Watch video

Gemini’s dual-chemistry architecture contains two cells using two different battery chemistries — each doing what they do best.
LFP for daily driving.
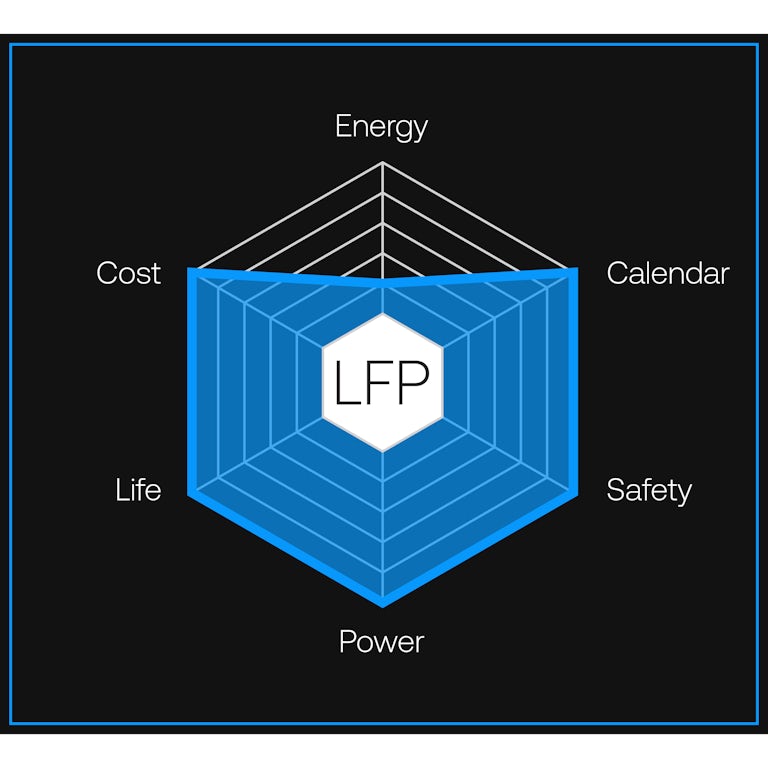
A lithium iron phosphate (LFP) cell powers the motor and meets the demands of daily driving. Gemini’s LFP cell delivers 441 Wh/L, which produces a range of 150 miles on a charge — more than enough power for 99% of trips.
Anode-free for longer trips.
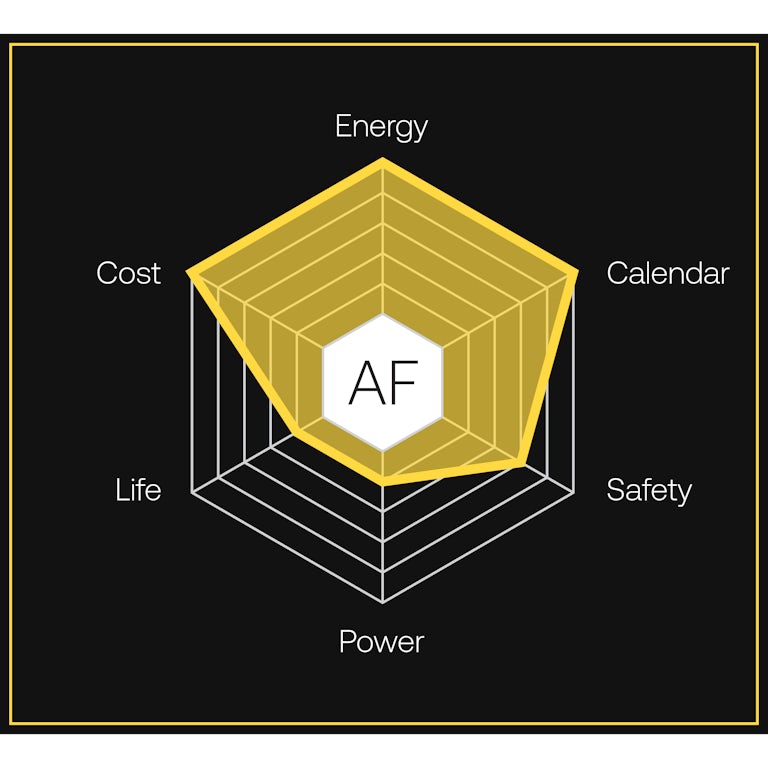
Gemini is road trip ready. After 150 miles, the anode-free cells begin powering the vehice. With an energy density of 1,007 Wh/L, the anode-free cells provide an additional 450 miles of range. Combined, the dual-chemistry architecture enables one of the longest-range EV packs on the market — delivering more than 600 miles on a single charge.
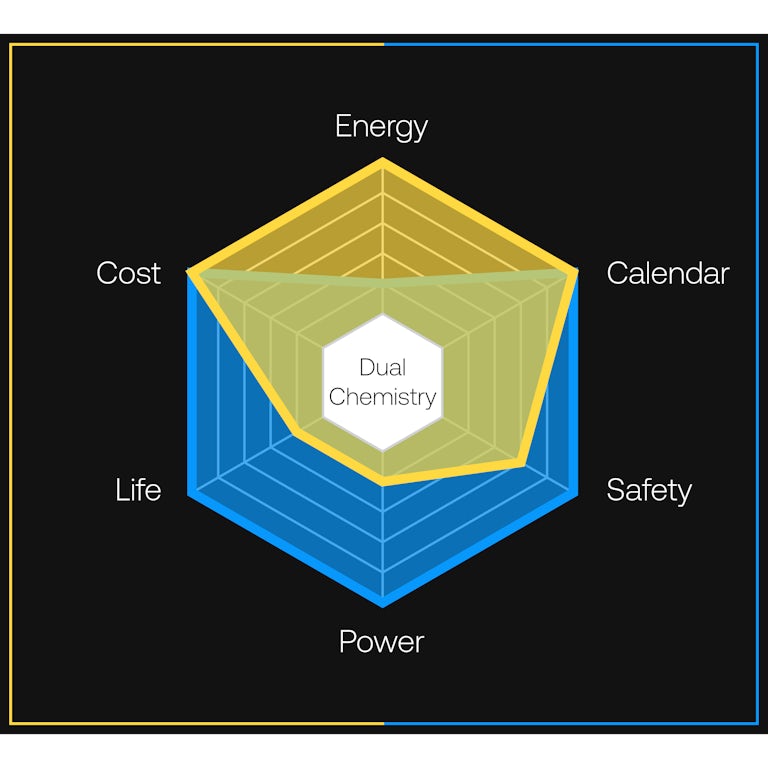
Gemini Dual-Chemistry.
Together, both chemistries deliver energy density and durability without compromise.More sustainable metals.
Gemini delivers more range than batteries that use nickel-and-cobalt-based chemistry. But Gemini maximizes materials that are abundant — iron for LFP, manganese for anode-free — while sharply reducing use of rare, expensive metals like nickel and cobalt.

Faster to market.
Gemini’s anode-free cell lowers costs by using abundant materials and a simpler production process. As a result, we can bring our advanced anode-free chemistry to market faster.
Gemini is safer, too.
By replacing nickel, cobalt and graphite with other materials, Gemini greatly reduces the risk of thermal runaway. And our patented skip-cell architecture helps prevent overheating in the pack.
Anode-free explained.
All batteries have an anode and a cathode. But battery makers also use the term “anode” to refer to the active material placed on the battery’s current collector during the manufacturing process. This material may contain graphite or silicon. We create our “anode” without this material. Instead, during “formation” — an essential step during which the battery is charged and discharged — small amounts of lithium plate onto the current collector. This process forms the anode in our anode-free battery.
Less material, lower cost.
Our advanced anode-free chemistry significantly lowers costs by using manganese-rich cathodes and no cobalt. Using less material allows us to fit more energy density into the cell, further increasing range.

Gemini Battery Powers BMW iX 608 Miles on a Single Charge
The Gemini dual chemistry battery achieved 608.1 miles of range in a BMW iX during a Worldwide Harmonized Light Vehicle Test Procedure (WLTP) test.
Subscribe to our newsletter
Let's talk about finding the right energy storage solution for you.

